|
Finally the theoretical calculation was done by using the calculation program Gaussian 03W, they are a permit to identify the phenomena engaged near the electrode and to completely determine the structures of the products of electrochemical oxidation formed during the degradation and which they are not quantifiable in experiments because of their high reactivity. 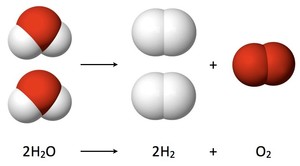
The BDD anode appears to be the more promising one for the effective electrochemical treatment of methidathion. For BDD the achieved Chemical Oxidation Demand reduction was about 85 %, while for SnO 2 it was about 73 %. The results provide that 2 % of NaCl, 60 mA cm −2 and 25 ✬ like the optimized values to carry out the treatment. Energetic cost has been determinate for all experiments. 
The influences of the electrode materials on methidathion degradation show that BDD is the best electrode material to oxidize this pesticide organophosphorous. ResultsĮlectrolysis parameters such as current density, temperature, supporting electrolyte (NaCl) have been optimized. The objective of this work is to study the electrochemical oxidation of aqueous wastes containing Methidathion using boron doped diamond thin-film electrodes and SnO 2, and to determine the calculated partial charge and frontier electron density parameters.  
Electrochemical oxidation considered to be among the best methods in waste water desalination and removing toxic metals and organic pesticides from wastewater like Methidathion.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
